A Snapshot of a Year in Progress
Between August 1, 2022, and July 31, 2023, FDA approved 14 new anticancer therapeutics, 12 previously approved therapeutics for treating new cancer types, and two new diagnostic imaging agents.
Home > AACR Cancer Progress Report > AACR Cancer Progress Report 2023: Contents
Between August 1, 2022, and July 31, 2023, FDA approved 14 new anticancer therapeutics, 12 previously approved therapeutics for treating new cancer types, and two new diagnostic imaging agents.

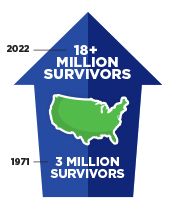
In the United States, the overall cancer death rate has been steadily declining since the 1990s, translating into more than 3.8 million cancer deaths avoided. However, progress has not been uniform against all cancer types, and there are stark inequities in the cancer burden among many sociodemographic groups.
Cancer is a collection of diseases that are characterized by unchecked cell multiplication. Both acquired and inherited genetic mutations, as well as modifications inside and outside the cell, can contribute to cancer development.
In the U.S., 40 percent of all cancers are associated with modifiable risk factors. The decline in cancer mortality over the past 30 years is largely attributable to reductions in smoking and increased screening.
Extensive research has shown that routine cancer screening saves lives. Advances in medical research are underscoring the potential for artificial intelligence and minimally invasive screening tests as new frontiers in early detection of cancer.

Advances in novel and innovative approaches to surgery, radiotherapy, chemotherapy, molecularly targeted therapy, and immunotherapy—the five pillars of cancer treatment—are saving and improving lives.

Cancer immunotherapeutics work by unleashing the power of a patient’s immune system to fight cancer and have emerged as one of the most exciting new approaches to cancer treatment. As of July 31, 2023, at least 23 different cancer immunotherapeutics have been approved by the FDA.
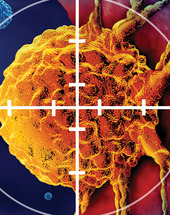
In this special section, Nobel Laureate James P. Allison, PhD, FAACR, and Padmanee Sharma, MD, PhD, discuss how bringing clinical trials, immune profiling, discovery science, data science, and drug development together on a coordinated team can accelerate the path of new drugs and drug combinations to the clinic.

Cancer survivors face unique challenges, but lifestyle factors like diet and exercise as well as caregiving resources like patient navigators, clinical care coordinators, and patient advocates can enhance patient outcomes.
Innovative technologies such as artificial intelligence and wearable devices, and novel approaches such as modulating the human microbiome, will drive progress against a wide range of cancers, including those that have been especially difficult to treat.

Federally funded research can accelerate the pace of new scientific breakthroughs against cancer, while policies and public health programs can ensure that the benefits of those advances are shared equitably.

This 13th edition of the Cancer Progress Report documents the unprecedented progress against cancer and underscores how continued investments in cancer research will ensure that treating and curing all cancers is within our reach.

The extraordinary advances against cancer detailed in this report were made possible by investment in biomedical research. To continue this progress, the AACR calls on Congress to provide robust, sustained, and predictable annual funding increases for NIH, NCI, FDA, and CDC in FY 2024 and beyond.

We are in an era of extraordinary scientific progress against cancer fueled by research-driven breakthroughs across the spectrum of cancer science. To maintain the momentum against cancer, we must ensure that medical research remains a national priority for our policymakers.

Unprecedented progress in medical research is increasing our understanding of the collection of diseases we call cancer and is driving remarkable improvements in cancer prevention, early detection, diagnosis, and treatment.

2023-2024 AACR President Philip D. Greenberg, MD, FAACR, serves as the chair of the Steering Committee for the AACR Cancer Progress Report 2023.
Your donation to the American Association for Cancer Research helps our more than 58,000 members worldwide drive progress against cancer.
Donate Today