Understanding How Cancer Develops
In this section, you will learn:
- Research provides our understanding of the biology of cancer, which is not one disease, but a collection of diseases characterized by the uncontrolled growth of cells.
- Genetic mutations underpin cancer initiation and development in most cases; the mutations are inherited in about 10 percent of cancer cases.
- Cancer initiation and progression are strongly influenced by interactions among cancer cells and cellular and molecular factors in their environment, referred to as the tumor microenvironment.
- The more we know about the contributions of the numerous individual factors and their interplay in influencing cancer development among all populations, the more precisely and effectively we can prevent and treat cancer.
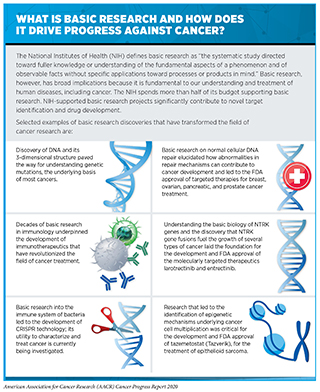
The extraordinary progress made against cancer as evidenced by the declining overall cancer death rate and the increasing number of survivors is a result of discoveries across the spectrum of cancer research from basic science to translational, clinical, and population research, which have deepened our understanding of how cancers arise and progress (see sidebar on What Is Basic Research and How Does It Drive Progress against Cancer?).
We now understand that cancer is a collection of diseases that arise when the processes which control normal cell growth, division, and life span go awry. As a result, cells start to multiply uncontrollably, fail to die, acquire unique ways to obtain nutrients for survival, and begin to accumulate. In body organs and tissues, the accumulating cancer cells form masses called tumors, whereas in the blood or bone marrow they crowd out normal cells. Over time, some cancer cells may invade distant tissues, a process termed metastasis, by entering the bloodstream or the lymphatic network, and form secondary tumors at remote sites.
Cancer Development: Influences Inside the Cell
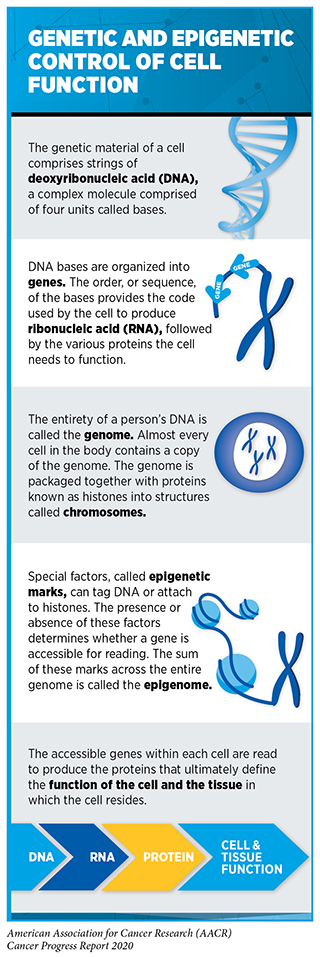
The normal behavior of each cell in the human body is controlled by its genetic material. The genetic material comprises chains of deoxyribonucleic acid (DNA), a complex molecule made up of four building blocks called bases. The four bases are organized in a very specific pattern to build two paired chains of the DNA that are packaged into condensed structures called chromosomes contained within a cell’s nucleus (see sidebar on Genetic and Epigenetic Control of Cell Function). Each person gets 23 chromosomes from each parent; thus, each normal cell has 46 chromosomes. The DNA is first converted into another complex molecule called ribonucleic acid (RNA) which is subsequently used by the cell to manufacture proteins. The order of the DNA bases and the way the DNA chains are packaged into chromosomes dictate which proteins and how much of them are made by each cell. Proteins are the molecules that perform important functions that dictate a cell’s fate.
Alterations in the DNA sequence, referred to as mutations, can disrupt normal protein function, and are the leading cause of cancer development (see sidebar on Genetic Mutations). Each person’s cancer has a unique combination of mutations, and as cancer cells divide, new mutations arise in the daughter cells. Thus, a tumor is made up of a collection of cancer cells with a wide range of genetic abnormalities. This variation in cell types, also known as heterogeneity, is an important part of a cancer’s characteristics and fuels the cancer’s ability to grow faster, escape therapy, evade the immune system, and metastasize to other organs.
While inherited genetic mutations play a role in about 10 percent of all cancer cases (see Table 2), most mutations are acquired over an individual’s lifetime due to errors arising during normal cell multiplication or because of environmental exposures, lifestyle factors, or coexisting health conditions (see sidebar on Sources of Genetic Mutations). Ongoing research continues to uncover new insights into the genetic basis of cancer (see sidebar on Unraveling the Complexities of Cancer Genomics).
Not all mutations acquired by a cell lead to cancer. In fact, the genes that are mutated, and the order and speed at which a cell acquires mutations, determine whether a cancer will develop and, if a cancer does develop, the length of time it will take to happen. The progressive nature of cancer provides distinct time points for medical intervention to prevent cancer, detect and/or intercept it early, and treat progressive disease. In general, the further a cancer has progressed, the harder it is to stop the chain of events that leads to the emergence of metastatic disease, which is the cause of most deaths from solid tumors (see Screening for Early Detection.
In addition to genetic mutations, changes in the physical structure of DNA caused by chemical modifications of the DNA and/or the proteins associated with it, termed epigenetic modifications, can lead to cancer development (see sidebar on Genetic and Epigenetic Control of Cell Function). Epigenetic modifications regulate how and when our genes are turned “on” or “off”, and they are made by specialized proteins that “add” or “erase” unique chemical modifications of DNA and/or histones (41). In contrast to genetic mutations, epigenetic changes are often reversible, providing an opportunity for therapeutic intervention. Our understanding of the role of epigenetics in cancer is, however, still incomplete, and continued research is needed to fulfil the real potential of the epigenome in cancer science and medicine. For example, according to a recent study, epigenetic alterations are associated with the development of certain forms of a rare cancer called gastrointestinal stromal tumors (GISTs) (42)WA F, Y D, SE J, ML H, DR T, E H, et al. Altered Chromosomal Topology Drives Oncogenic Programs in SDH-deficient GISTs. Nature [Internet]. Nature; 2019 [cited 2020 Jun 30];575.. More specifically, epigenetic marks called DNA methylation can displace certain anchors from the DNA. Loss of such anchors can lead to the activation of “on switches” in cancer-causing genes leading to tumor development.
Research aimed at the identification of genetic and epigenetic alterations that drive cancer development has led to the development of a new class of therapeutics—molecularly targeted therapeutics—which aim to rectify the cellular changes that arise due to such alterations. While these advances have revolutionized cancer treatment, they have also brought attention to the fact that individuals of European ancestry are grossly overrepresented in most clinical research investigations (43)(44)Sirugo G, Williams SM, Tishkoff SA. The Missing Diversity in Human Genetic Studies. Cell [Internet]. Elsevier Inc.; 2019;177:26–31.. The lack of racial and ethnic diversity in human genomic studies limits our understanding of cancer biology, including inherited cancer predisposition, in underrepresented populations. Rectifying this issue is an area of active research investigation, as reported in the AACR Cancer Disparities Progress Report 2020 (14).
Cancer Development: Influences Outside the Cell
Cancer arises due to the disruption of normal cellular functions through genetic and epigenetic changes in a cell. Once cancer is initiated, however, complex interactions between cancer cells and their surrounding environment—known as the tumor microenvironment—contribute to disease progression.
The tumor microenvironment is a specialized niche surrounding the cancer cells in a tumor and consists of immune cells—components of one’s natural defense mechanism—as well as other cellular and molecular elements (see sidebar on Cancer Growth: Local and Global Influences). Bidirectional communications between cancer cells and their microenvironment affect tumor growth and metastasis (45)Mcgranahan N, Swanton C. Review Clonal Heterogeneity and Tumor Evolution : Past , Present , and the Future. Cell [Internet]. Elsevier Inc.; 2017;168:613–28.(46). For instance, recent studies show that an important function of the local microenvironment is to provide sources of energy that promote multiplication and/or survival of metastatic cancer cells in an otherwise unfavorable environment (47)Zeng Q, Michael IP, Zhang P, Saghafinia S, Knott G, Jiao W, et al. Synaptic proximity enables NMDAR signalling to promote brain metastasis. Nature [Internet]. Nature Publishing Group; 2019 [cited 2020 Jun 30];573:526–31.(48)Y Z, A W, N C, CE P, K X, GM A, et al. Polyunsaturated Fatty Acids From Astrocytes Activate PPARγ Signaling in Cancer Cells to Promote Brain Metastasis. Cancer Discov [Internet]. Cancer Discov; 2019 [cited 2020 Jun 30];9.(49)A T, B F, V R, JM U, B S, A S, et al. Metabolic Heterogeneity Confers Differences in Melanoma Metastatic Potential. Nature [Internet]. Nature; 2020 [cited 2020 Jun 30];577.. The tumor microenvironment can also shelter cancer cells from the effects of radiation, chemotherapy, and immunotherapy, thereby rendering them resistant to treatment (50)Sun Y. Tumor microenvironment and cancer therapy resistance. Cancer Lett [Internet]. Elsevier Ireland Ltd; 2016;380:205–15..
Future studies that uncover additional cellular and molecular properties of the tumor microenvironment will be vital for improving cancer diagnosis and treatment. In this regard, a series of recent reports characterized the cellular and molecular landscape of lung cancer cells and associated immune and other tumor-supporting cells in the microenvironment, across different regions in the tumor and over time (51)Swanton C. Take lessons from cancer evolution to the clinic. Nature [Internet]. Nature Publishing Group; 2020 [cited 2020 Jun 30];581:382–3.. The data provide deep insights into the mechanisms by which the immune microenvironment interacts with lung cancer cells and vice versa in different regions within a tumor and across different tumors in a patient (52)AbdulJabbar K, Raza SEA, Rosenthal R, Jamal-Hanjani M, Veeriah S, Akarca A, et al. Geospatial immune variability illuminates differential evolution of lung adenocarcinoma. Nat Med [Internet]. Nature Publishing Group; 2020 [cited 2020 Jun 30];1–9.(53)Joshi K, de Massy MR, Ismail M, Reading JL, Uddin I, Woolston A, et al. Spatial heterogeneity of the T cell receptor repertoire reflects the mutational landscape in lung cancer. Nat Med [Internet]. Nature Publishing Group; 2019 [cited 2020 Jun 30];25:1549–59.. Furthermore, the studies characterize the wide range of dynamic alterations including mutations within immune and cancer cells that enables lung cancers to evade attack and elimination by the immune system (54)Ghorani E, Reading JL, Henry JY, Massy MR de, Rosenthal R, Turati V, et al. The T cell differentiation landscape is shaped by tumour mutations in lung cancer. Nat Cancer [Internet]. Nature Publishing Group; 2020 [cited 2020 Jun 30];1:546–61.(55)Rosenthal R, Cadieux EL, Salgado R, Bakir M Al, Moore DA, Hiley CT, et al. Neoantigen-directed immune escape in lung cancer evolution. Nature [Internet]. Nature Publishing Group; 2019 [cited 2020 Jun 30];567:479–85.(56)López S, Lim EL, Horswell S, Haase K, Huebner A, Dietzen M, et al. Interplay between whole-genome doubling and the accumulation of deleterious alterations in cancer evolution. Nat Genet [Internet]. Nature Publishing Group; 2020 [cited 2020 Jun 30];52:283–93.. These discoveries have critical implications in understanding cancer progression and relapse, as well as response to state-of-the-art treatments such as immunotherapies.
Cancer Development: Integrating Our Knowledge
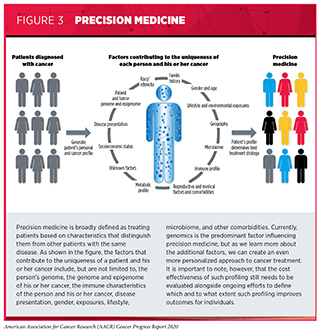
Over the past decade, we have made significant progress in how we understand and treat the complex group of diseases we call cancer. We have learned that cancer development is influenced by many factors including a patient’s biological characteristics, social and environmental exposures, and lifestyle. Therefore, each person’s cancer is unique, and as a result we are beginning to see a major shift from a “one size fits all” paradigm to cancer prevention, screening, and treatment to a more personalized approach called precision medicine (see Figure 3). The aim of precision medicine is to use information about a person’s genes, proteins, and environment to prevent, diagnose, and treat disease.
Precision medicine aims to use genetic and other information about a patient’s tumor, as well as other factors, to help diagnose, plan treatment, determine how well treatment is working, or make a prognosis, with the overarching goal of improving clinical outcomes and minimizing unnecessary diagnostic and therapeutic interventions. While genomics is the predominant factor currently guiding this approach, adoption of precision medicine has already shown substantial benefits in survival in patients with cancer (58)Sicklick JK, Kato S, Okamura R, Schwaederle M, Hahn ME, Williams CB, et al. Molecular profiling of cancer patients enables personalized combination therapy: the I-PREDICT study. Nat Med [Internet]. Nature Publishing Group; 2019 [cited 2019 Jun 19];25:744–50.(59)Pishvaian MJ, Blais EM, Brody JR, Lyons E, DeArbeloa P, Hendifar A, et al. Overall survival in patients with pancreatic cancer receiving matched therapies following molecular profiling: a retrospective analysis of the Know Your Tumor registry trial. Lancet Oncol [Internet]. Elsevier; 2020 [cited 2020 Jun 30];21:508–18.. Precision medicine holds tremendous potential in cancer science and medicine, given that we are experiencing a rapid evolution of state-of-the-art technologies that are enabling researchers to uncover additional key players in cancer development, such as novel tumor genomics, immune system, energy metabolism, lifestyle, and environmental exposures, at an unprecedented pace. An area of active focus is the accumulation of relevant data from racial and ethnic minorities, the lack of which really minimizes the current implementation of precision medicine for these populations (60)Creed JH, Berglund AE, Rounbehler RJ, Awasthi S, Cleveland JL, Park JY, et al. Commercial Gene Expression Tests for Prostate Cancer Prognosis Provide Paradoxical Estimates of Race-Specific Risk. Cancer Epidemiol Biomarkers Prev [Internet]. Cancer Epidemiol Biomarkers Prev; 2020 [cited 2020 Aug 1];29:246–53.. Going forward, concerted efforts from all stakeholders in medical research will be critical in order to deliver the full potential of precision medicine to the entire cancer community.